Published on LinkedIn: https://www.linkedin.com/pulse/when-ok-cheat-win-business-oliver-canty



“We have evidence that warnings were ignored and that these products continued to be used despite the warnings and that following the infections…[there was] a cover-up,” – Andy Evans, chairman of campaign group Tainted Blood
Speaking to the BBC, Mrs May said: “They deserve answers, and the inquiry that I have announced today will give them those answers, so they will know why this happened, how it happened”
These quotations from today’s news refer not to the Deprox, but to the tragic suffering and loss of life from infected blood administered by the NHS in the 70s and 80s. At last, there is to be an inquiry, and the victims’ relatives will know how and why 2,400 NHS patients died.
Meanwhile a disturbingly similar tragedy is unfolding right here and now – and again the NHS turns a blind eye for fear of negligence lawsuits, and the corrupt medical suppliers hastily reap the profits before the mounting evidence of fraud and corruption forces them back to the dark places that spawned them.
The victims – the frail elderly, the cancer patients, the cystic fibrosis sufferers, have no idea that the C. difficile or MRSA infections they have suffered from were preventable. They are assured that the hospital rooms and equipment have been sterilised by the best technology available – the Deprox – which according to the Hygiene Solutions Ltd. website, “…achieves a log 6 reduction of even the most virulent of organisms.”
The truth is in stark contrast to this glib fabrication; – leaked emails and the testimony of former Hygiene Solutions employees prove that the Deprox units are turned up to their maximum output of RH30 when tested, but in everyday use are turned down to output levels of RH 5 to RH 15 – with directly proportional reductions in H2O2 concentration and germicidal efficacy.
To compound this error, a recent letter published in the Journal of Hospital Infection revealed that most of the tested efficacy of the Deprox process could be attributed to the exceptionally high silver nitrate content of the Deproxin solution – an illegal additive which Hygiene Solutions now claims to have removed.
What residual efficacy remains absent this powerful cytotoxin remains untested and unproven.
The shocking and highly persuasive statistics from the UCLH hospitals, showing a substantial upwards step change in C. difficile rates corresponding exactly with the years of Deprox use, cannot be denied. These are public data sets, and the 75 or so extra infections over this period demand explanation.
We need an inquiry in to the Deprox scandal NOW, while lives can still be saved – not in 30 years time.
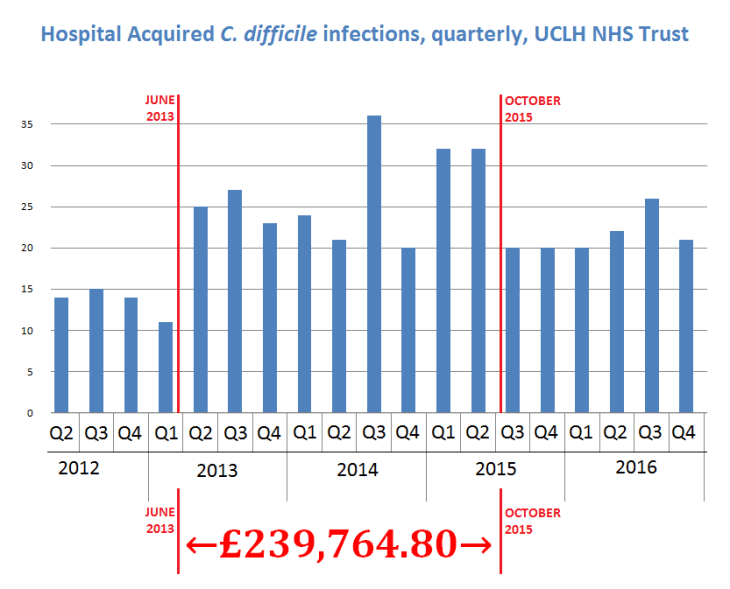
A Freedom of Information request¹ to UCLH disclosed the starting and finishing dates of their disastrous Deprox decontamination contract with Hygiene Solutions Ltd. The Deprox operations started in June 2013 and ran continuously, 7 days per week through to October 2016. The contract called for at least 4 Deprox units to be at the hospital, and 6 or more processes to be completed daily.
However, due to frequent breakdowns, Hygiene Solutions struggled to meet their obligations, and on occasion asked engineers to put a non-functional Deprox unit in a room, tape up the door and “run” a process – thus not only defrauding the NHS but leaving a dangerously contaminated room that the staff believed to have been sterilized.
Plotting the contract dates against the quarterly UCLH C. difficile data² (extended through 2016 with mandatory government reporting data)³ reveals an exact correlation between the period of Deprox deployment and a substantial step change in the number of C.difficile infections – approximately 70 extra cases over the 29 month period.
According to the March 2016 government report on C. difficile mortality, the 30 day mortality rates for the London area were about 17%. – suggesting that approximately 12 deaths in this period could be attributed to Deprox operations. Any patients who acquired C. difficile or any other Heathcare Associated Infection, (HAI) in the UCLH hospitals between June 2013 and October 2015 should consider contacting a medical negligence solicitor and seeking compensation.
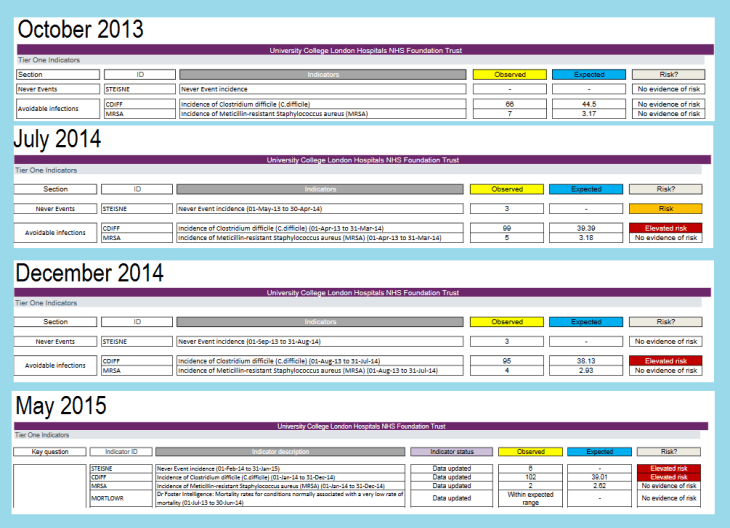
The CQC (Care Quality Commission) makes regular evaluations of NHS trusts using a list of critical indicators. These are the Intelligent Monitoring reports. The extracts from a series of these reports below show how the “Incidence of C. difficile” indicator moved from “No evidence of risk” to “Elevated Risk” when the Deprox program was implemented. The complete reports can be found at: http://www.cqc.org.uk/provider/RRV/reports
[1] https://www.uclh.nhs.uk/aboutus/wwd/Annual%20reviews%20plans%20and%20reports%20archive/Infection%20Control%20Annual%20Report%202015-16.pdf (See graph, p.17)
[3] https://www.uclh.nhs.uk/aboutus/FOI/FOI%20disclosure%20list/FOI2017271Response.pdf
https://www.dropbox.com/s/71u3j3fcdqwqfnj/ResponseFOI2017271.xlsx%20%28~13%20KB%29.URL?dl=0

Judge Adele Williams
Judgement was pronounced on a £26,000 NHS fraud last week. The perpetrators only escaped jail as they had pleaded guilty at the earliest opportunity and had repaid the money in full.
But Judge Adele Williams described it as a
She said further: –
What would the Judge have to say to the perpetrators of an £8,000,000 fraud against the NHS? Even if the directors of Hygiene Solutions were to refund the NHS for the tens of thousands of fraudulent Deprox processes they have done over the years, (see https://deproxfraud.info/2017/03/13/leaked-emails-prove-test-cheating-bodily-harm-and-massive-fraud/ ) would they escape jail?

Hygiene Solutions claim a single Deprox unit has the capacity to decontaminate rooms with a volume of 380m3, e.g. a 12 bed ward bay. A typical hospital side ward (single room with ensuite) has a volume of 60m3.
However, the chamber used by Hygiene Solutions Ltd to test the Deprox is 1.5m x 1.5m x 2.8m. Total volume 6.3m3 , just 10% of the volume of a hospital single bedroom, and 1.7% of the maximum volume that Deprox is guaranteed to disinfect. It is barely larger than a telephone box.
Hygiene Solutions internal testing, published here for the first time reveals that the Deprox, in spite of being boosted with 50% more concentrated hydrogen peroxide solution than the standard “Deproxin” was incapable of a log 6 decontamination of even this tiny test chamber.
The Deprox was thoroughly tested over a period of months by David Sempere Aracil, a well qualified chemist. David placed Log 6 biological indicator discs (Apex Biological Indicator #HMV-091) in 8 different locations around the inside walls of the test chamber. The Deprox unit (“Trusted by leading hospitals around the world”) was sealed in the chamber, and the process was run. The log6 BIs were incubated – they were all alive.
David tried substituting Sanosil SO15 which at 7.5% H2O2 is 50% more concentrated than Deproxin. Now some of the BIs would be sterilised, sometimes. Over several weeks in late 2014, David did a series of 12 tests in the test chamber, all with 7.5% H2O2 rather than the 5% Deproxin. He tried turning the ΔRH up and down, but to no avail. In 5 of these tests, all 8 BIs remained viable. None of the tests sterilised more than 6 out of 8.
In summary then:
Deprox, running on a 5% H2O2 solution, is claimed to give a log6 decontamination of an entire 380m3 ward, including inside small crevices and complex equipment. In Hygiene Solutions’ own tests, the Deprox running on a 7.5% solution, and thus generating a 50% higher aerial H2O2 concentration than the standard process, completely failed to give a log6 decontamination of a 6.3m3 box in multiple tests.
Hygiene Solutions continued to promote and sell the Deprox with exactly the same claims, but in 2015, they turned the whole Deprox fleet down from ΔRH20 to ΔRH5. See https://deproxfraud.info/2017/03/13/leaked-emails-prove-test-cheating-bodily-harm-and-massive-fraud/
Fortunately, (or unfortunately for Hygiene Solutions) David’s notes of these tests survived.
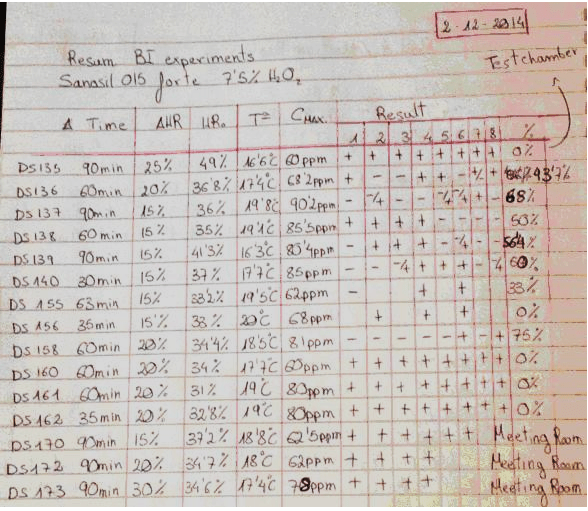
This table is a summary of 15 tests done by David Sempere Aracil, assisted by Tautvydas Karitonas, over a period of months. Both are university graduates with extensive research experience, and David has a PhD in Chemistry. The tests were done with a standard production model Deprox machine, the purpose of the tests was to determine if the extremely low efficacy of the Deprox process could be rectified by increasing the concentration of the hydrogen peroxide solution from the standard 5% to 7.5%.
The results were recorded in 3 A4 hardcover notebooks. Each of the 15 tests was recorded in more detail on preceding pages of the notebook. In addition to the efficacy tests, the notebooks contain extensive details of tests on prototype catalyst systems, and constitute proof that HS was well aware of both the low efficacy and residual gas issues with Deprox.
Heading: “Sanosil 015 forte” refers to Sanosil S015, which is a disinfectant intended for water systems. It is 7.5% Hydrogen peroxide solution. Note that this is more concentrated than the 5% Deproxin solution that is used in production Deprox machines.
Col.1. The test number. These are not sequential, as some tests did not use Biological indicators (BIs) and were not recorded in this resume.
Col. 2 Duration of test measured from when the machine starts vapourising. (It takes several minutes for the machine to fill the piezo tank at the beginning of each test)
Col. 3 Delta HR setting of machine. This is adjusted by using unmarked pressure sensitive switches below the LCD display. – see How to test your Deprox.
Col. 4 HRO This is the original relative humidity in the test chamber before the machine starts.
Col. 5 TO Temperature in the chamber before the machine starts
Col. 6 CMAX Hydrogen peroxide concentration in PPM, maximum level reached during process.
Results columns. The first 12 tests were done in the test chamber (wardrobe). Each number represents a specific marked location on the test chamber wall where an exposed stainless steel Bacillus subtilis log6 biological indicator was placed. The chamber is a crude wood and plasterboard structure in an essentially unheated warehouse. It is approximately 5’ x 5’ x 9’ and the indicators were placed at various heights on the interior walls of the chamber. The last 3 tests were done in the company board room which is approximately 12’ x 25’.
A” +” indicates that the BI still contained viable bacteria, a “–“ indicates that all bacteria on this indicator were killed.
Final column. This is the percentage of BIs that were killed.

The Yeovil Hospital Norovirus outbreak, first announced on April 3rd is now in its 21st day, and has spread from the initial 2 affected wards to a third..
Incredibly, the hospital decided to attempt to halt this highly contagious form of gastroenteritis WITHOUT using bleach or disinfectant, instead putting their trust in the Ultra-V device from Hygiene Solutions, which has never been tested on norovirus or any other type of virus before.
In fact, the only published study of Ultra-V shows that it has an efficacy of LESS THAN LOG 1. This test was done in the Wye Valley NHS Trust hospital in Hertford, and the sampling and incubation of the samples was carried out by Hygiene Solutions themselves.
To put that in context, ordinary household Dettol, as everyone knows, kills 99.9% of germs. The Ultra-V by the manufacturer’s own test results, kills less than 90%, and that is only where the UV light hits the surface directly. In the shadowed areas of the room it will be almost completely useless.
So in real terms, if a door handle was carrying 1000 norovirus germs, ordinary household Dettol would leave on average one germ alive. Ultra-V would leave more than 100 germs alive, and only if the light could somehow illuminate both sides of the handle at once.
Since 1865, when Joseph Lister introduced carbolic acid as a wound dressing, and operating theatre spray, countless millions or perhaps billions of lives have been saved by simple disinfectants, both in healthcare settings and by household disinfectants and chlorinated drinking water. There is probably no other single discovery that has done more to relieve human misery and suffering. 150 years of experience, scientific testing and advances in chemistry have produced an excellent range of effective, proven disinfectants, that are both inexpensive and can be applied by very simple spray and wipe methods.
Why then should all this be abandoned, at the worst possible moment, in favour of a hugely expensive, unproven device that by the manufacturer’s own published test results achieves only 100th of the effectiveness of ordinary Dettol?

There has been much contention about the relative merits of vapour and fog based “HPV” systems, but the most important difference has been overlooked, which is that crude fogging systems such a Deprox will only reach on average 50% of the surfaces in a treated area. The effect has been masked, perhaps deliberately in some cases by the testing protocol, that always places BIs in the most exposed areas and favourable orientations.
In order to understand the problem, we need to shed some light on the difference between fogging and vapour based systems:
Definition of vapour
The scientific definition of a vapour is a gas, as opposed to an aerosol or fog. As a general rule, fogs and aerosols scatter light, and are visible as a white cloud. A gas or vapour is non-scattering, hence invisible.
How do vapour systems work?
Vapour phase systems emit invisible gaseous hydrogen peroxide into the air, and maintain the concentration at a high level (>100ppm) for about an hour, which will give a 6 log efficacy against most pathogens. The vapour is generated thermally from a 30% hydrogen peroxide solution.
How do fogging systems work?
Fogging systems emit an aerosol of small (5-10 micron) droplets of low concentration hydrogen peroxide solution (typically 5%). There are two different mechanisms of disinfection occurring simultaneously:
Vapour concentration is limited by Henry’s law to an average over the process time of about 50ppm. Hence these systems are capable of a log 4 efficacy, provided that the starting humidity of the room is low enough to allow the droplets to evaporate.
Homogeneity
Hydrogen peroxide is a “lazy gas”. Its high molecular mass gives it a slow rate of diffusion, hence auxiliary fans should always be used to ensure the gas is thoroughly mixed and distributed throughout the room to be treated. If this is done, a vapour system will give a homogeneous distribution of gas, which will disinfect all surfaces regardless of orientation or distance from the generator. The H2O2 molecules break down continuously, with a half life of about 50 minutes, so if left to diffuse naturally, the concentration will drop substantially with distance from the machine.
Fog Shadowing
Fogging systems have altogether different dynamics. The fog droplets fall continuously under gravity relative to the surrounding air. For example, a 2 micron fog droplet will fall by 50 times its diameter per second. This has a dramatic effect on the distribution of the active ingredient on the surfaces in the room. In order to wet a surface, and thus transfer the H2O2 to the pathogens present, the droplets have to impinge on it for sufficient time and with sufficient force to break the surface tension. For upwards facing horizontal surfaces, the gravitational settlement is adequate, and these surfaces will have a visible film of moisture at the end of a process. A horizontal downwards facing surface, such as the underside of a table or door handle will remain dry.
Unless the airflow direction is varied, i.e. by multiple oscillating fans, droplet contact will still be extremely uneven, with almost all the drops impacting on the side of the object facing the airflow. This is perfectly illustrated by this picture of a rime frost. Here a combination of a supercooled fog and a light breeze has caused the droplets to impact and freeze on the side of the fence wire facing the wind. The opposite side of each wire has no ice at all. This is very similar to the shadowing effect of the Ultra-V systems – on average, 50% of surfaces are “shadowed” and hence untreated.

Disinfection of walls and other vertical surfaces is a lottery. The degree of wetting, and thus disinfection is at the whim of the air currents in the room, and droplets impinging on the wall at a shallow angle will bounce, particularly if the surface is at all hydrophobic.
None of this would matter if the vapour level was adequate, as the vapour would disinfect the surfaces not wetted by the droplets. However, in humid conditions, the vapour level is greatly reduced – leading to a very patchy and inadequate performance.
Temperature and circulation
The emission from fogging systems is cold – as much as 15 degrees cooler than the room temperature, due to the cooling effect of droplet evaporation. This cold dense vapour falls rapidly to floor level. The effect can be very visible on starting machines that eject the vapour vertically – the cloud of fog will often not reach the ceiling before collapsing and flowing down to the floor. In the case of the Deprox, the air inlet is on the bottom of the machine, so creating a circulation cell where the fog rises through the machine in the centre of the room and in the rest of the area is moving downwards.
 The effect of this is to give heavy droplet wetting on the upwards facing surfaces around the machine. Bed rails, for example will be wet on the top and dry on the bottom. This has the fortuitous (for the manufacturer) effect, that if the machine is tested in the customary manner by placing biological indicators around the room in petri dishes facing upwards these samples will be exposed to a substantial “rain” of droplets and may show high efficacy levels which are not at all representative of the real disinfection achieved. If the BIs were secured in a vertical or downwards facing orientation, much lower efficacies would be recorded.
The effect of this is to give heavy droplet wetting on the upwards facing surfaces around the machine. Bed rails, for example will be wet on the top and dry on the bottom. This has the fortuitous (for the manufacturer) effect, that if the machine is tested in the customary manner by placing biological indicators around the room in petri dishes facing upwards these samples will be exposed to a substantial “rain” of droplets and may show high efficacy levels which are not at all representative of the real disinfection achieved. If the BIs were secured in a vertical or downwards facing orientation, much lower efficacies would be recorded.
Perhaps the best way to determine the real efficacy of a fogging system in a typical side ward application is to attach the BIs to the walls of the ensuite. This is usually the area most distant from the machine, and also the most contaminated. Attaching the BIs to the walls in a vertical orientation will remove the effects of falling droplets.
Effect of humidity
Because both types of system are evaporating an aqueous solution, the humidity in the room rises during the process as water is evaporated into the air along with the H2O2.
The more concentrated 30% solution used by true vapour systems obviously means that less water is evaporated, and these systems can achieve the target aerial concentrations even in conditions of high initial humidity.
By contrast, the fogging systems using a 5% solution must evaporate 19 units of water for each unit of H2O2. They cannot function at all over about 70% starting humidity, and in anything but the driest weather conditions, the level of disinfection achieved will be limited. This is because the evaporation of the initial outflow of droplets quickly raises the humidity to saturation, and fog emitted later in the cycle cannot evaporate, remaining as a highly visible white cloud.
Most fogging systems claim to be a “dry mist” and are set to avoid condensation, as this can damage electronics and stain furnishings. The only way to avoid condensation is to turn the machine off when the humidity reaches 90% or so. Thus while the Deprox has a fixed cycle time, in humid weather, firstly much of the H2O2 is locked up in the fog droplets, and secondly much less solution used by the system, as it will spend most of the cycle “waiting” for the humidity to drop to a level where its control system will restart the fog generation. (Remember that the H2O2 is continually breaking down, so without continuous replacement, the concentration drops rapidly.)
Even this automatic regulation seems to have been inadequate, as former employees of Hygiene Solutions report that they were told to turn down the units to very low levels at times of high humidity to avoid problems.
This is not a minor effect – Deprox users will have noticed how on some days almost no fog is visible, while on others there is a dense fog which is still visible at the end of the “deactivation” cycle. It is also unsurprising that it is the systems in south Wales that have been suspended – the high humidity in this area leads to frequent process fails and residual fog.
Finally…
There are now fogging systems on the market that use electrostatics to give an even distribution of droplet wetting regardless of orientation. This is a technology that has been used in paint and agricultural chemical spraying for decades, and causes the fog to “wrap” around objects and coat all faces evenly. I have no data as to the efficacy of this process, but it is a promising idea.
More detailed data about the differences between fogging and vapour systems can be found at https://www.linkedin.com/pulse/h2o2-fog-vapour-efficacy-tests-examined-richard-marsh?published=t
A major Welsh health board, with more than ten hospitals has suspended both Deprox and Ultra-V in all of its sites. The suspension is in response to numerous incidents of healthcare workers being exposed to high levels of toxic Deproxin fumes on re-entering a room after decontamination. Affected staff have been given a precautionary medical examination.
The RH (i.e. the amount of hydrogen peroxide in the air) of these machines was set to 13, where according to the manufacturer, a level of RH 40 is needed to give a log6 disinfection. This would account for reports of persistent contamination in some units in spite of repeated Deprox use. (You can easily find the RH setting of your Deprox here.)
Even with the machines turned down to almost a quarter of the required concentration, the level of H2O2 at the end of the process was so high that in many cases there was still a clearly visible fog in the air – and these incidents occurred AFTER Hygiene Solutions had fitted a catalyst to all its Deprox units in response to the serious accident in the Royal Worcester Hospital. This is further proof that the catalyst is just a placebo, to hide the fact that the ONLY way to ensure a safe atmosphere after a Deprox process is to turn down the H2O2 levels to 1/8th of the level needed for disinfection.
A similar problem was encountered by the Cwm Taf University Health Board, which covers the North Glamorgan area. They were sent a Deprox that had accidentally been set to RH 20, leaving the ward filled with a choking fog of chemicals after the “green light” had illuminated, indicating that the room was safe to enter. The Health Board used gas detector guns to measure residual hydrogen peroxide left after the process and it was 15 times the safe limit.
It should be pointed out that the Deprox machine has NO MEANS WHATEVER of monitoring the deactivation process. The green light is simply on a 45 minute timer, and will illuminate at this time regardless of the level of gas in the room.
Hydrogen peroxide gas detectors are expensive to buy, but can be rented at very reasonable rates from Drager UK. The best detector for this application is the Drager X-am 5100. The contact number for gas detection enquiries is 01670 352891.
Note: Do NOT rely on on gas detectors supplied from Hygiene Solutions – these are calibrated to show only a fraction of the real gas level! This can very easily be demonstrated by using a HS supplied unit and a rented Drager unit side by side.
In the face of mounting evidence as to the grave danger this process poses to both healthcare workers and patients, it would seem a wise move to discontinue the use of this equipment until the official HSE enquiry has made its recommendations.

Move over Wikileaks – now we have Ricki-leaks…
A large batch of very interesting internal Hygiene Solutions emails have fortuitously come into my possession. Too much material for one post so this will just be the first of three…

In January 2015, Mark Fentiman sent an urgent message that all Deprox units were to be turned down to RH5. Now it emerges that this order was in response to the incident above, in which a healthcare worker at the Royal Worcester Hospital inhaled Deproxin fumes and suffered serious respiratory problems. (Note that ISS Healthcare operate the Deprox machines as a subcontractor in some NHS hospitals.)

The Log6 claim so loudly and insistently trumpeted by Hygiene Solutions is based on a machine set to RH40. The RH setting gives the amount of H2O2 per cubic metre of air, so resetting the machine to RH5 only gives 12% of the required concentration. In reality, the machine is just a placebo – no useful or meaningful level of disinfection is possible at this level.
The shocking fact is that Hygiene Solutions continued to provide their Deprox systems and “decontamination” service at this dangerous and utterly ineffective level, did not inform their 60 or so NHS Hospital customers of the change, and continued to charge full price, a tidy sum of £2,500,000 per year.
Given that approximately 5000 NHS patients die each year from the very infections that this system is supposed to prevent, there can be no doubt that this action led to completely avoidable infections and death, as well as robbing the NHS of millions of pounds in fees for thousands of imaginary decontamination services that did not actually take place.
To follow:
