It’s been dubbed the “diesel dupe”. In September, the Environmental Protection Agency (EPA) found that many VW cars being sold in America had a “defeat device” – or software – in diesel engines that could detect when they were being tested, changing the performance accordingly to improve results. The German car giant has since admitted cheating emissions tests in the US.
BBC News
The following article was written in order to expose the egregious, multi-million pound fraud perpetrated by Specialist Hygiene Solutions Ltd on the NHS. The company directors have created a tangled web of lies and misinformation in which to securely conduct their criminal activities. One of their most devious and effective tactics is to conduct rigged tests of their equipment, with the specific intent of impressing influential medical researchers and practitioners, whose credentials are then hijacked to promote the product.
The product in question is Deprox, a hydrogen peroxide vapour generating device that is claimed to effect a one million fold reduction (log6) in bacteria and viruses, in hospital wards, intensive care units and operating theatres. In fact, in extensive tests the reduction in pathogen numbers was found to be less than log2, which is an extraordinary 10,000 fold less than the performance claimed.
Hygiene Solutions uses the same ploy as VW to conceal the deficiencies of their Deprox process. Former employees report that it is standard practice to turn up the hydrogen peroxide (H2O2) levels by 50% on machines that are undergoing biological indicator trials at hospitals, and to turn them down by 75% where hospitals are using H2O2 meters to check that the gas had reduced to safe levels before re-entering the room.
Hygiene Solutions are thus very cagey about trials taking place outside of their knowledge or control, hence in the “Bolton NHS Trust Method Statement, 2014” we have the following:
Hygiene Solutions will agree with suitably qualified persons before commencing the process the preferred method of validation. Recommended methods include:
• Surface sampling before and after for specific organisms – subject to prior agreement with suitably qualified persons regarding surfaces, location, organisms and laboratory protocol. Details will be recorded onto the Validation Record and corresponding Floor Plan for future reference and audit.
• Surface seeding with sampling before and afterwards for specific organisms – subject to prior agreement with suitably qualified persons regarding surface seeding, location, organisms….
Etc…
The key phrase here is “prior agreement”. This allows time for a Hygiene Solutions technician to access the machine in question and turn it up or down according to whether a biological or gas detection trial is planned. As more and more hospitals are using Deprox, this juggling game has become more difficult to sustain. In at least one case, a hospital used a H2O2 meter without permission and found dangerously high gas levels on re-entering the room. Hygiene Solutions explained to them that this was a false reading, because “The gas monitor had not been calibrated to the Deprox machine…”
The obvious question is, what if a hospital wants to test the gas levels and biological indicators simultaneously? There is a second line of defence – Hygiene Solutions can claim that the gas levels used in the process are “proprietary information” and insist that they are not measured. This is a common practice across most HPV equipment manufacturers – few published studies of HPV decontamination contain any measurements of the H2O2 concentration in the air during the process.
These clandestine adjustments to the treatment parameters clearly have a substantial impact on both the safety and efficacy of the process. For the sake of clarity, I have addressed the issue of the Deprox being turned down in a separate report, see “Deprox and the Emperor’s new clothes”.
Now we will look at evidence that Hygiene Solutions has “duped” leading academics at UCLH in order to obtain a highly creditable peer reviewed paper validating their claimed efficacy for the Deprox process.
Turning up the Deprox vapouriser has its limitations. Even at full power, the Deprox process is incapable of reaching anywhere close to the claimed log6 efficacy. Hence there are no published, peer reviewed papers to support this claim. Hygiene Solutions have temporarily staved off the problem by stating that tests have been done to validate this claim, and are “awaiting publication” .
Here is an example from the Sheffield Response to Tender 2014
YES, the Deprox decontamination system has been independently validated to be effective against Mycobacteria in the environment. Hygiene Solutions are currently awaiting the publishing of a study to this effect. Carried out in a clinical setting, this work was based around pre-seeded plates with known quantities of specific organisms placed at a variety of locations around the room to prove the homogeneous diffusion of the H2O2 vapour produced by the Deprox system, in both the main patient room and the en-suite bathroom. Due to the peer review and publication process, these studies are not in the public domain currently but should be available under NDA (non-disclosure agreement) directly between Sheffield Teaching Hospitals NHS Foundation Trust and the publishing parties.
Precisely the same mantra is repeated for other pathogens for which Sheffield required data. It should be taken in to consideration that Mycobacteria is catalase positive and hydrophobic – making the organism one of the hardest to inactivate using H2O2 vapour. It is now two years since this tender was submitted and no such report has been published. These claims clearly become less and less credible as time passes.
By 2015 Hygiene Solutions had a serious problem. They rightly considered the log6 claim as essential to their marketing strategy, but knew it would not stand up to critical examination. Both medical professionals and competing business were on the verge of uncovering the truth behind the extraordinary claims made for Deprox, and at least two “whistleblowing” attempts had been made by former workers. What could the company do to avoid exposure and keep the lucrative contracts on upwards of 150 Deprox units around the UK and Australia?
In January 2016 Hygiene Solutions seemed to have a lucky break:
S. Ali et al. (University College London Hospital) published a paper apparently proving that the 5% Deprox system could produce a log6 decontamination, and had performance indistinguishable from a much more expensive system (Bioquell Q10) that was using 30% H2O2 solution.
In conclusion, S. Ali et al. say:
“Both systems demonstrated similar turnaround times (2–2.5h), and no differences were observed in the efficacy of the two systems against BIs…”
And
“The starting concentration and mode of delivery of hydrogen peroxide may not improve the efficacy of decontamination in practice…”
A truly extraordinary result! However, as astronomer Carl Sagan famously said:
“Extraordinary claims require extraordinary evidence.”
Therefore, these conclusions can hardly be expected to pass without question. We will now review the evidence presented for these claims.
It is obvious and fundamental that the efficacy of a disinfectant in solution is proportional to its concentration. A 5% hypochlorite solution will be much less efficacious than a 30% hypochlorite solution, and the difference will be entirely reproducible. In fact, if the concentration of a hypochlorite solution is unknown, it can be accurately determined by measuring its efficacy against known BIs.
According to the paper:
- Both systems tested were using the same reagent (H2O2 vapour)
- Both systems produced remarkably similar results against a whole range of pathogens and soiling levels.
- Both had similar process times.
It is inescapable therefore that both systems generated very similar aerial concentrations of H2O2. The “mode of generation” is irrelevant. H2O2 vapour has precisely the same properties regardless of the means by which it has been evaporated, and of the concentration of aqueous solution from which it was derived.
The questions now become :
- What was the H2O2 vapour concentration?
- Is it possible to generate this from a 5% solution?
We can find the H2O2 vapour concentration needed from the following paper:
The inactivation of Bacillus subtilis spores at low concentrations of H2O2 vapour
D J Malik et al.
http://www.sciencedirect.com/science/article/pii/S0260877412004141
This paper plots the log disinfection values of H2O2 vapour for concentrations from 50ppm to over 3000ppm, on bacterial spores. From the data and equations given, we derive the following graph:
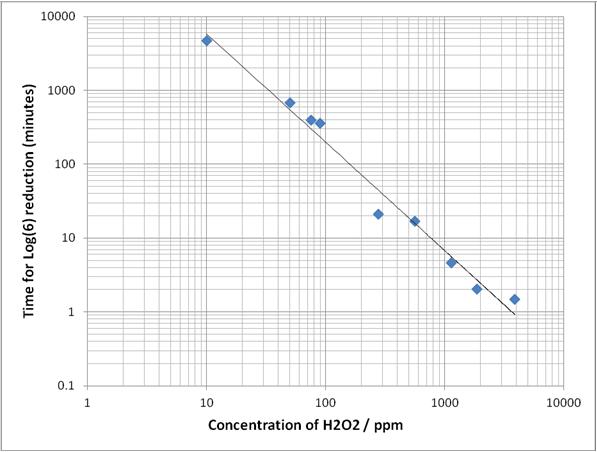
The above graph is derived from the data fit using the Weibull model described in Malik et. al., Journal of Food Engineering 114 2013) 391-396
Given that the log reduction results over a wide range of BIs was practically identical for both the Deprox and the Bioquell system, and the process times were similar, we can say with confidence that both systems generated similar average H2O2 vapour concentrations in the tests, and from published data that this concentration was in the region of 170ppm.
Is it possible for Deprox to generate and sustain this concentration of H2O2 in the air for 90 minutes?
To answer this we need to understand how the Deprox evaporation mechanism works. The Deprox has an “atomisation chamber” being an open topped stainless steel tank with piezoelectric transducers in the bottom face. In operation the level of solution in the chamber is kept at a constant depth of about 50mm. The transducers produce ultrasonic vibrations which propagate upwards to the top surface of the solution, where they vibrate the air/liquid interface so energetically that it is broken up in to a fog of tiny droplets. These droplets have the same composition as the bulk solution. The fog of droplets is drawn out of the chamber and ejected vertically in to the air by a fan. All this can be seen from the patent.
The effect of this “atomisation” is to greatly increase the surface area of solution exposed to the air in the room, hence to increase the rate of evaporation proportionately.
Deprox is specifically marketed as a “dry” process. The disinfection is not due to the droplets wetting the surfaces. (Although as they fall by gravity, they do wet horizontal, upward facing surfaces.) The disinfection is due to the H2O2 gas released when the droplets evaporate in the air.
The room to be treated is sealed, and there is a very large surface area of solution distributed around the room as a fog of droplets. Also, the air in the room is being repeatedly circulated through the atomisation chamber. It is obvious therefore that the H2O2 in the air will quickly reach an equilibrium with the H2O2 in solution, the aerial concentration being dependant on the aqueous concentration. (Henry’s law)
From published data, we can find the equilibrium concentrations of H2O2 gas over a range of solution concentrations as below:
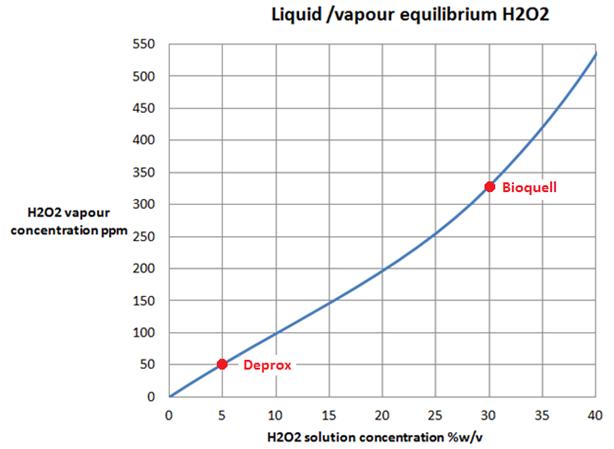
The equilibrium concentration of H2O2 over a 5% aqueous solution is about 50ppm. Thus if a Deprox machine was to run in a room that already had a concentration of 100ppm H2O2 in its atmosphere, the effect of the fog of droplets would be to remove H2O2 from the air and concentrate it in the solution, until the equilibrium was restored.
Clearly then, it is impossible for a simple process like Deprox, that merely exposes a large area of dilute solution at ambient temperature and pressure, to sustain aerial concentrations of H2O2 in excess of the equilibrium value, i.e. approximately 50ppm.
In order to sustain the required 170ppm of gas in the air, the Deprox machine would require at least a 17% H2O2 initial solution.
We can see this equilibrium effect in practice by looking at the graph below from the paper “Disinfection Using Hydrogen Peroxide” by TNO Built Environment and Geosciences (This is a paper distributed by Hygiene Solutions to potential customers to support the Deprox performance claims.) This graph shows how the aerial H2O2 concentration develops during a Deprox type process using a5% solution. After an initial peak of 105ppm, the H2O2 level rapidly drops to the 50% equilibrium line.
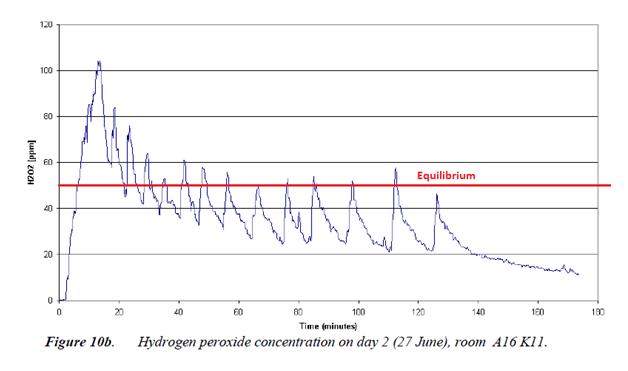
It may be asked how the initial peak is attained if the process is limited by the equilibrium effect. The explanation is that the first pulse of droplets emitted from the machine in to the dry air evaporate completely, releasing all of their H2O2 in the process. At this point there is no large surface area of 5% solution in the room to absorb the excess. Visually, white fog can be seen rising from the machine and disappearing as it evaporates.
After a few minutes the relative humidity in the room reaches saturation, and the droplets can no longer evaporate, but remain in the air in increasing numbers. Visually the room now appears to be filled with white fog. The droplets, all at 5% concentration, now absorb the excess gas and maintain the aerial concentration at equilibrium.
There is another reason why a 5% solution cannot produce 170ppm of vapour, which is as follows:
From fig.10b above we know that by evaporating a 5%H2O2 solution we can reach a momentary peak of 100ppm H2O2 in the air, but this rapidly drops off, giving an average over 90 minutes of about 50ppm. In order to increase the H2O2 concentration from 50ppm to 170ppm, we need to inject 3.4 times more H2O2 into the atmosphere of the room, by vapourising 3.4 times as much of the solution. This inevitably also requires 3.4 times more water to be vapourised in order to release the H2O2 in gas phase.
This is clearly impossible, as the RH quickly reaches saturation, and the solution will no longer evaporate. In other words, a 170ppm aerial H2O2 concentration would require an impossible relative humidity of around 340%.
Another severe limitation of the Deprox is that it does not have a dehumidifier. Competing atomisation based systems use a dehumidifier to dry the air prior to starting evaporation. This allows much more H2O2 to be introduced in to the room in the initial atomisation pulse before the RH reaches saturation.
Humidity in a UK hospital room can be up 70%RH if the room has just been wet cleaned or if the weather is humid. As Deprox is marketed as a dry, no condensation process, the atomisation is set to cut off once 90%RH has been reached. In order that the process will still work in humid weather, and because the Deprox has no dehumidifier, the machines are set up to only give a 20% rise in RH (70 + 20 = 90) This setting is called “ΔRH” i.e. change in relative humidity. The way this works can be seen from the patent:

Fig. 10 b above was for a machine that was set to ΔRH40. The production Deprox machines are set to ΔRH20, so would not actually produce even the 50ppm average concentration seen on this graph. The volume of solution evaporated by a production Deprox would be half that shown on the graph, so requiring an even more concentrated solution if it is to reach the required 170ppm.
We can do an approximate estimate of this as below.
At 5% solution and ΔRH40, the “atomisation” process as used by Deprox gives an average aerial concentration of 50ppm over 90 minutes.
Therefore at 5% solution and ΔRH20, the aerial concentration would be around 25ppm.
To increase this to the required figure of 170ppm, the concentration of the initial solution will need to be 6.8 times greater – 6.8 x 5 = 34%.
From this estimate, we must consider the possibility that the Bioquell and Deprox machines were actually using very similar initial concentrations of solution (30 – 34%) and thus achieved comparable results. This intuitively sounds more likely than S. Ali’s conclusion “Although the starting concentration of hydrogen peroxide used in HPS1 was higher (30%) than in HPS2 (4.9%) (Table III), there was no significant difference in the efficacy of the two HPV systems…”
There is further support for this from the table below:
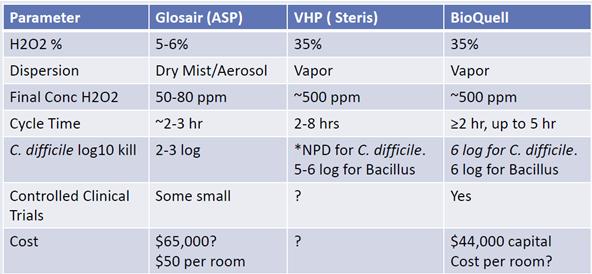 This is from a comparative study by Dick Zoutman, MD, FRCPC Emeritus Professor, Medical Microbiology & Infectious Diseases, Queen’s University.
This is from a comparative study by Dick Zoutman, MD, FRCPC Emeritus Professor, Medical Microbiology & Infectious Diseases, Queen’s University.
The report compares the Glosair 5% solution atomisation based system, (similar to Deprox but has a dehumidifier), with two other systems that use a 35% solution. The 35% systems both achieve log5-6 and the 5% system achieves log2-3. The Glosair system is a sophisticated and long established machine with a dehumidifier incorporated. The company is owned by Johnson & Johnson and thus has very substantial R & D resources. It is highly improbable that the Deprox, using the same 5% solution, and the same atomisation based process, can achieve a result that is 3log higher ( i.e. 1000 times better) than Glosair.
All the scientific evidence points to an average aerial concentration of H2O2 for both machines of at least 170ppm, and an initial solution concentration in the region of 30ppm. Why then do S. Ali et al. claim a 5% initial concentration, which by Henry’s Law fundamentally limits the sustained aerial concentration to 50ppm? What evidence do they give of this fundamental parameter? None whatever – they have simply repeated the information given to them by the machine’s manufacturer.
We must now ask, is Hygiene Solutions a credible source for this key information?
Examination of the Hygiene Solutions sales literature reveals numerous contradictory, misleading and downright dishonest statements, all with the aim of making the Deprox process appear much better than it really is. A small selection of these (39 to be exact) is given on the home page of the website www.deproxfraud.info. Two of these statements, which can be independently verified, bear repeating here:
Cambridge graduate microbiologist Rick Fentiman, the company’s technical director, said: “There is a lot of evidence to support the fact it can reduce outbreaks by half the time it takes to manage an outbreak using traditional methods.
http://www.worcesternews.co.uk/news/10026455.A_new_weapon_in_the_war_on_germs/
This is untrue – Rick Fentiman is NOT a microbiologist, and has no university qualifications whatever.
The Deprox system is designed, manufactured and tested to exacting British and International standards, including ISO 14001, ISO13485, ISO9001 and is CE marked. (Sheffield Teaching Hospitals Response to Tender, 2014)
Fraudulent – Hygiene Solutions does not have ISO13485 accreditation. (ISO13485 is the ISO standard for medical equipment manufacture)
Needless to say, a company that will lie shamelessly about ISO accreditation and university qualifications, would hardly hesitate to misrepresent the concentration of a chemical should there be a compelling commercial reason to do so. Viewed from a commercial perspective, it is obvious that the paper published by S.Ali et al gives a very compelling case to hospitals to purchase the Deprox system rather than the much more expensive and inconvenient Bioquell system. It also validates their much repeated but never proven log6 claim, which was coming under increasing criticism from both potential customers and from competitors. It is interesting also that prior to the publication of S.Ali’s paper, Hygiene Solutions never published the “log6” claim on their website – it was only made in sales literature and tender submissions that were not in the public domain. Within a few days of the paper being published, the Hygiene Solutions website had the following oddly ungrammatical statement prominently displayed:

See http://hygiene-solutions.co.uk/deprox/
How much then is Deprox worth to Hygiene Solutions, to justify such desperate measures?
Deprox accounts for practically all of Hygiene Solutions’ revenues, as their only other product line, Ultra-V, has only just been launched. The revenues from Deprox can be estimated from the prices quoted on the tender submissions, as below;
There are now approximately 160 Deprox systems in 60 hospitals throughout the UK plus a few systems in Australia. The weekly rental cost of the system for a 36 month contract is £1196/year plus £680 to renew the contract every three years. The basic level of “support cover” which gives the required service every 200 processes costs £1250/year. The 2 litre refills have a list price of £78.30 each, and last for “2 or 3 processes”.
If we assume that each system is used 2 or 3 times a day, we get the following annual expenditures by Deprox users:
Deproxin refills……… £28,579
Rental ……………. £1422
“Bronze support cover” £1250
TOTAL approx £31,250 per system per year.
Multiply this by 160 systems in operation and the total annual revenues garnered by Deprox come to just over £5,000,000.
This is all the more impressive when we consider that most of this sum comes from the Deproxin refills at £78.50 for a 2 litre bottle. A 5 litre bottle of 6% hydrogen peroxide solution can be purchased in the UK for £1.64 + VAT – i.e. 33p/litre. The approx 1g/litre of silver nitrate is worth about 40p, so the total cost of a litre of Deproxin is about 70p.
For a business that operates out of a few spare rooms in the back of an agricultural sundries warehouse, and has no visible external signage to advertise its existence, these are remarkable profit margins and revenues.
The UCLH tests by S.Ali et al. presented an existential threat to Hygiene Solutions. There is abundant evidence that atomisation based systems using 5% solution will achieve log3 at best against bacterial spores. Had the tests given a log3 reduction for Deprox as compared to a log6 result for Bioquell, the result would have been devastating for Hygiene Solutions, as all of its existing contracts are based on the assurance of a log 6 reduction, and Bioquell and Johnson & Johnson (manufacturers of the Glosair system) would have no doubt publicised this result widely, leading to potential lawsuits from existing customers, and the elimination of Deprox from the market as a viable product.
The directors of Hygiene Solutions had a stark choice. Leave the 5% “Deproxin” in the test unit and face financial ruin and humiliating exposure as fraudsters, or simply substitute a 30% solution, humiliate the competition, and open the door to ongoing and increasing revenues of millions of pounds per year.
How could this substitution have been effected? Witnesses confirm that a standard, sealed Deproxin refill bottle was inserted in to the machine prior to the test, clearly labelled as 4.9% hydrogen peroxide.
There is a hint as to the likely modus operandi in the “Specifications” page at the end of the Deprox instruction manual:
Fluid storage capacity: 10 litres (full), 13 litres (max)
The Deproxin refill bottles are only 2 litre, so the Deprox must have a substantial internal storage reservoir for the working solution. This can be seen from the drawing in the patent, as below:
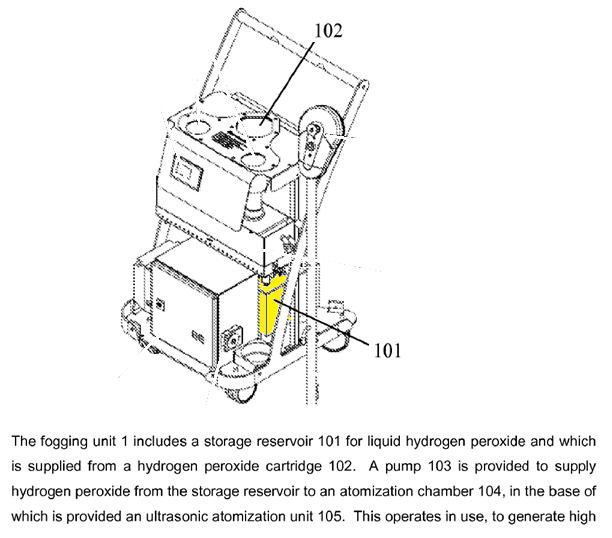
This 10 litre storage reservoir is only accessible by removing the cover of the machine and is sealed by solenoid valves except when the pump is in operation or it is being topped up from the Deproxin bottle inserted at 102.
Clearly this tank could be pre-filled with a high concentration H2O2 solution before the test, and the addition of a 2 litre 5% refill would not cause significant dilution.
It is interesting also that S. Ali et al. comments in his paper that
“…during this study, both parties provided storage of equipment and hydrogen peroxide stock solutions off-site.”
The machine tested then was not a standard unit taken from the hospital’s fleet, but a machine brought in especially for the test.
Hygiene Solutions could have felt quite confident that the substitution would not be detected, as if the UCLH team had decided to check the solution concentrations, they would have doubtless taken a sample from the refill bottle provided. It is probable that they were not even aware of the existence of a large storage tank within the machine.
In conclusion:
- The directors of Hygiene Solution had very compelling financial and reputational reasons to cheat in this test.
- They had a history of dishonest and fraudulent claims.
- They had an easy way to make a substitution.
- They had good reason to believe that this substitution would be unlikely to be discovered.
On the assumption that Henry’s Law has not been repealed, the only other possible explanation for the anomalous test result is human error in the execution of the tests. This has been suggested by Dr Jonathon Otter of Imperial College, see;
https://reflectionsipc.com/2016/04/01/diluting-the-efficacy-of-hydrogen-peroxide-room-decontamination/
However, given the extensive practical experience and high reputation of Prof Peter Wilson who oversaw the tests, and the excellent laboratory facilities of the University College of London Centre for Clinical Microbiology, this can be dismissed. (see http://www.ucl.ac.uk/infection-immunity/research/res_ccm/#)
If Hygiene Solutions wish to dispute the claim that they cheated in this test, then they should allow a repeat test where a sample of the Deproxin is taken from the internal storage tank and tested for concentration. I am quite certain that they will be very reluctant to do so.