News May 16th 2017
32,000 views – Volunteers needed!

Deproxfraud.info celebrates 32,000 views this month. Numerous former Hygiene Solutions employees have made contact, as have NHS, Australian and New Zealand healthcare personnel, epidemiologists, academic microbiologists, and directors and staff of honest and conscientious healthcare equipment manufacturers. Many of these businesses have lost tens to hundreds of thousands of pounds worth of contracts to the flagrant dishonesty and bald faced lies used by Hygiene Solutions in their tender submissions.
The volume of material accumulated is substantial, and continues to rise. Incredibly, Hygiene Solutions have not learned from their mistakes and are now attempting to promote their Ultra-V using the same deceitful practices as they used for the now defunct Deprox. Drowning men clutch at straws…
More news is already in the pipeline. The HSE is conducting a thorough investigation of the company, its systems and its products. Their report, when published, will be of great interest to deproxfraud.info subscribers. A number of “freedom of information” requests have been put out and the results will be found here very soon. Ongoing correspondence is maintained with MPs, members of the House of Lords, NHS Protect officials and Trades Union representatives.
Since the whistleblower site was launched at the Infection Prevention 2016 Harrogate Conference I have put thousands of hours and no small amount of funds in to maintaining and expanding this website. Results have been deeply gratifying – Hygiene Solutions have been stopped in their tracks, with no updates at all on their website since the exposure began, and no new sales of the Deprox system whatever. .
Ultimately, all these actions will result in lives being saved and a substantial reduction in HAI incidence across the UK, as hospitals replace these fraudulent products with effective systems from honest and competent manufacturers.
However, I think it is time to move on, so I am looking for volunteers among the many beneficiaries of the website to take over the editorial role. I recognize that the new editor or editors may prefer to be anonymous, and also that it may be best for the role to be carried by several people rather than one individual. It will be good also to get a wider variety of views and opinions, so I will be looking for guest blogs to be published, alongside the substantial body of draft articles that I will be passing on to my successors.
Please contact me at richard.marsh@romer-photonics.com, or via my facebook page https://www.facebook.com/richard.marsh.921230 if you are interested in being a part of the deproxfraud.info team!
Specialist Hygiene Solutions Australia Pty Ltd, the sponsors for the Deprox and Deproxin products in Australia has sunk. The ABN has been cancelled, and the company no longer exists. There are thought to be about 20 units in Australia, as follows:
- Westmead Hospital, Sydney: 2 Deprox
- Royal North Shore Hospital, Sydney: 2 Deprox
- Concord hospital, Sydney: 2 Deprox
- Mater Private Hospital, Brisbane: 6 Deprox
- 8-10 units in storage in the SafeSmart Access building, Ermington, NSW
As Deprox has no Australian sponsor, they can no longer be legally used.
Sources inside the UK manufacturer, Specialist Hygiene Solutions Ltd, say that the Australian and New Zealand machines are only serviced on an “ad hoc” basis as there are limited servicing facilities in Australasia. Certainly the service records show that some of these machines have not been serviced for over a year, (see Deprox Fleet Report last 3 pages). This will cause poor fogging performance and increasing lead oxide contamination as the piezo discs erode, see https://deproxfraud.info/toxic-cocktail-lead-and-silver-nitrate/
News 2nd February
New webpage – all the data and videos re. lead and silver nitrate in Deproxin. Click on this link Toxic Cocktail – Lead and Silver Nitrate
News 26th January

Specialist Hygiene Solutions Australia Pty Ltd has applied for voluntary liquidation. The company is the AU “sponsor” for the UK manufactured Deprox HPV bio-decontamination systems, i.e. it has full legal liability for the product in Australia. The liquidation application comes within days of the commencement of the HSE investigation in to Specialist Hygiene Solutions Ltd, the UK manufacturer.
“Sponsor” in this context means “A person taking responsibility for the actions of another” and it is a requirement for any company exporting medical equipment to Australia to have an Australian resident, or an Australian owned corporation to sponsor the equipment and register it with the Australian Government Theraputic Goods Administration. (TGA)
The responsibilities of the sponsor are extensive, and include mandatory reporting of the following to the TGA –
Any adverse events or problems with the products.
Any malfunction or deterioration in the characteristics or performance of the kind of device; or
Any inadequacy in the design, manufacture, labelling,instructions for use or advertising materials of the kind of device;
– that have led to any complaint or problem in relation to the kind of device, no matter how minor.
The full regulations run to 331 pages and can be downloaded here:
https://www.tga.gov.au/publication/australian-regulatory-guidelines-medical-devices-argmd
The sponsor is responsible for “Post-market vigilance and monitoring requirements” so liquidation of the sponsor requires withdrawal of the registered devices and products from use. The liquidation takes effect on February 2nd 2017.
The matter is being investigated by the Australian Government Department of Health Therapeutic Goods Administration, under the reference number below:
DEVICE INCIDENT REPORT DIR 44353 – Deproxin
News 2nd November
- Whistleblower site http://www.deproxfraud.info has had 14,000 hits as of this morning!
Deprox PbO (lead oxide) dust emissions. Are operators protected?
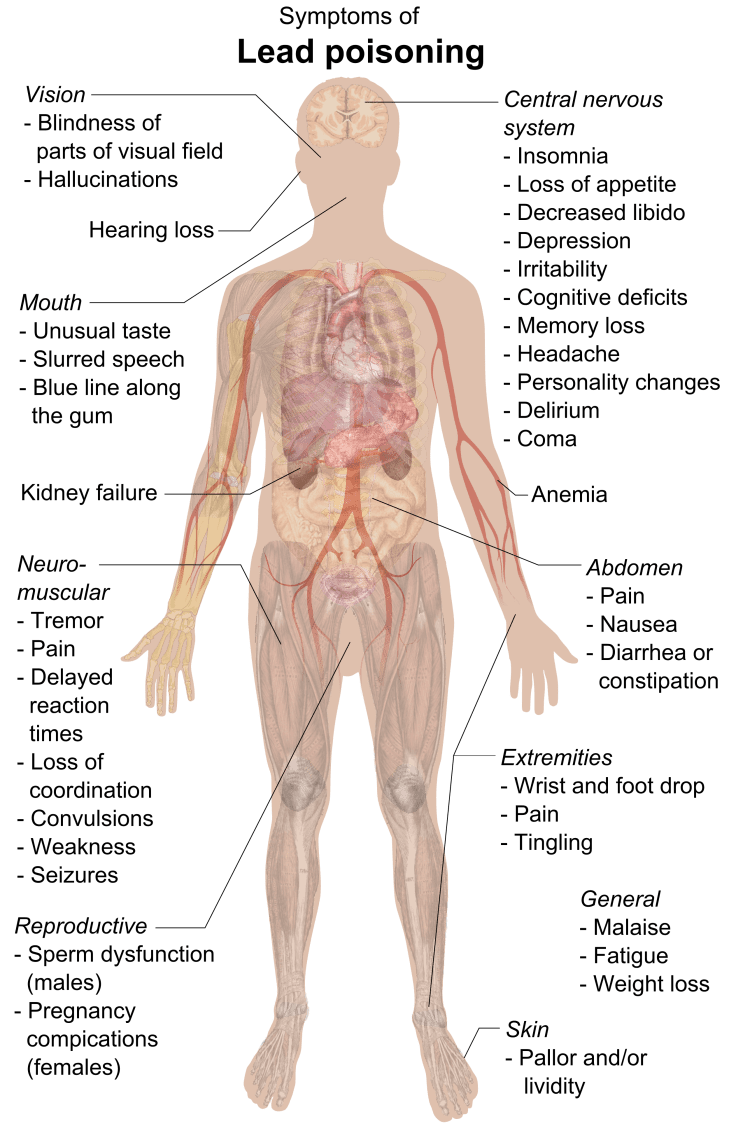
- This photo shows crusts of toxic lead oxide on the piezo disc of a Deprox machine. There are 8 of these discs in each Deprox and they are 70% lead. See “Lead zirconate titanate”. As this corrodes away, the lead oxide mixes with the Deproxin and is emitted with the fog as a fine powder – thus ultimately ending up in the lungs and internal organs of Deprox operatives. Lead is a cumulative poison, damaging the brain and reproductive system.

Graphic by
News 31st October
Operatives Exposed to Toxic Chemical at 2000 x the Legal Limit
Deproxin, the chemical solution fogged in to the air during the Deprox process, contains 4g of toxic silver nitrate per bottle. (That is enough to kill a child if ingested). As the fog droplets of solution evaporate to release the H2O2 vapour, the silver nitrate crystallises out as a black dust, which can clearly be seen where it has settled on the top of the machine and other white exposed surfaces after the process is complete.
Because it is particularly damaging to the lungs, the legal limit for exposure to airborne silver nitrate dust is very low – 0.01mg/m³ – that’s 1/100,000th of a gram per cubic metre of air.
A typical hospital side ward is 60m³, and uses about 1/3 of a bottle of Deproxin. The process therefore puts a total of about 1.33g of silver nitrate in to the room, which comes to 22mg/m³. This is 2200 times in excess of the legal limit. The hydrogen peroxide in the air breaks down naturally or is (in theory) removed by the catalyst. However, the silver nitrate does not break down, is NOT removed by the catalyst, and the airflow of the “deactivation unit” will keep the dust suspended in the air, rather than allowing it to settle. The first person to re-enter the room at the end of the process is the Deprox Operative, who not only breathes in the highest concentrations of dust before the room is ventilated, but is exposed to this multiple times every working day.
Hygiene Solutions directors try to hide this serious health hazard from their empolyees by mislabelling both the Deproxin bottles, and falsifying the official Deproxin MSDS (Health and safety data sheet) Both state the active ingredient as “Silver 0.2%” The MSDS however has to give the correct reference number for the chemical, which is CAS: 7761-88-8. This is the number for silver nitrate – NOT silver. Silver (inert metal) has no more in common with silver nitrate (toxic oxidising compound) than sodium chloride (table salt) has in common with chlorine (poisonous green gas). To call silver nitrate “silver” is not a little white lie, it is a big dirty dangerous lie.
Operatives should NOT be reentering the room without full face respirators. All exposed surfaces should be wiped down with wet disposable wipes to remove accumulated dust, and an appropriate meter for silver nitrate should be used to check the air quality before nursing staff or patients are readmitted.
If you have any questions, Mr Jit Patel, Hygiene Solutions Health and Safety Officer, will be able to look up CAS: 7761-88-8 and provide appropriate equipment.
Symptoms of silver nitrate inhalation are:
| Sore throat. Cough. Burning sensation. Shortness of breath. Laboured breathing. Blue lips or finger nails. Blue skin. Dizziness. Headache. Nausea. Confusion. Convulsions. Unconsciousness. Symptoms may be delayed. |
Silver compounds are not excreted, they accummulate permanently in the organs of the body, including the brain, skin and eyes. This can cause a condition called Argyria, where the whites of the eyes and the skin become permanently stained a blue-grey colour.
What are the clinical features of argyria?
Whether or not a person exposed to prolonged or high levels of silver develops argyria depends on many factors including the dose and form of exposure, the duration of exposure, and the route of exposure (ie: ingested, inhaled, skin contact). What is apparent is that the degree of hyperpigmentation in patients is directly correlated with the amount of sliver present.
Generalised argyria usually begins with a grey-brown staining of the gums which later progresses to involve large areas of the skin.
- Over a period of months or years depending on the degree of exposure the skin turns a bluish-grey, metallic, to slate-grey colour.
- The hyperpigmentation is most pronounced in sun-exposed areas such as the face (particularly forehead and nose), neck, arms, and hands. Hence, the discolouration is thought to result from the presence of silver and silver-induced increase in melanin concentration.
- The fingernails, conjunctival membranes and mucous membranes may become hyperpigmented.
- In some cases, the entire skin turns a slate blue-grey colour.
There is currently no effective treatment or cure for Argyria.
News 26th October
Tim Murrell speaks out!
Tim Murrell, former Technical Manager at Hygiene Solutions Ltd, named as “Inventor” on the Deprox patent, has condemned both the process and the lies and dishonesty used to sell it to the NHS. Tim referred to the 39 dishonest and misleading statements quoted on the deproxfraud.info page “A Tangled Web” and confirmed that all of these claims made about Deprox were indeed false. Tim recalled challenging director Rick Fentiman about the claim that Deprox had built-in leak detection and would shut down if a door was opened. Rick refused to remove the claim saying “That’s marketing.”
Tim was aware that if he remained an employee he could be held legally responsible as a collaborator if the fraud was exposed. He was so concerned about being the “Technical Manager” of such an unscrupulous business that he left the company’s employment after only a year and from then on only did electronic design for them on a self-employed consultancy basis. Tim gave a full report last week to the Norfolk CID concerning the Deprox fraud.
If you have information about Deprox, please call 101 and ask for DC 36 872 FLITNEY.
News 22nd October
Long Prison Sentences for NHS Fraudsters
In a case of medical equipment fraud comparable to the Deprox scandal, the perpetrators received prison sentences in excess of 5 years. This fraud only related to a sum of £250,000 and there was no threat to the health or lives of any NHS patients. (see account below) The directors of Hygiene Solutions and their accomplices will face MUCH longer sentences than these when convicted, as the Deprox fraud has taken an estimated £6,000,000 from the NHS and has also caused much avoidable suffering and almost certainly fatalities to NHS patients.
Three sentenced for £250,000 NHS pressure chamber fraud. Two men who ran a private decompression chamber designed to treat potentially fatal cases of “the bends” in divers and their accomplice were sentenced today for conspiring to defraud the NHS of £250,000. Their sentencing at Plymouth Crown Court concludes a major investigation by the NHS Counter Fraud Service and Devon and Cornwall Police. David Welsh was jailed for five years and three months. Michael Brass received two years and three months, in his absence. Anthony Robert Walker received a two year suspended sentence. Former boss of the Fort Bovisand Hyperbaric Diver Centre David Welsh, 51, of Pomphlett Road, Plymstock, colluded with diving instructor Michael Brass, 45, of Liverpool and Walker to charge the NHS for very expensive emergency decompression treatments that never took place. They used the names, addresses, dates of birth and national insurance numbers of people who had never suffered from “the bends” or been treated in the company’s hyperbaric chamber. The case centred on bogus invoices submitted to 12 health bodies relating to 37 “patients” supposedly treated between 1998 and 2002. Managing Director of the NHS Counter Fraud Service Dermid McCausland, said: “This was not a victimless crime. For more than four years, these men systematically and shamelessly diverted resources intended for patients requiring emergency decompression treatment, simply to enrich themselves. The taxpayer picked up the bill of £6,500 for every treatment these men fraudulently claimed to have given. We are now working with the police to pursue compensation orders to get back every penny for the NHS.” Devon and Cornwall Constabulary said: “This was a complex case that necessitated close liaison between the investigating bodies. That co-operation led to this successful prosecution and acts as a warning to those who believe taxpayer’s money is an easy target. You will be caught. You will be prosecuted.”
News 21st October
Thank You!
The response to the appeal for witnesses was immediate and hugely significant. On Thursday I was a lonely whistleblower struggling against the odds to persuade the CID to take the (admittedly almost unbelievable) story of the Deprox fraud seriously. Now several ex-employees of Hygiene Solutions Ltd, including personnel involved from the start in the design and testing of Deprox, have come forward and FULLY and INDEPENDENTLY confirmed the wretched and despicable fraud perpetrated against the NHS by these crooks from their King’s Lynn warehouse.
News 20th October
Norfolk Constabulary opens Fraud Investigation.
The CID opened the case yesterday, and has already collected from me a substantial body of evidence, including documents, electronic data and a fraudulently mislabeled Deproxin bottle.
I am appealing for witnesses in connection with this issue. If you have any information pertaining to this matter, please call Crimestoppers anonymously on 0800 555111 or through its anonymous online form at http://www.crimestoppers-uk.org.
If you are concerned about the possible consequences to yourself of reporting fraudulent activity, please read the section below, or go to www.gov.uk/whistleblowing/what-is-a-whistleblower from which this section is copied.
What is a whistleblower
You’re a whistleblower if you’re a worker and you report certain types of wrongdoing. This will usually be something you’ve seen at work – though not always.
The wrongdoing you disclose must be in the public interest. This means it must affect others, eg the general public.
As a whistleblower you’re protected by law – you shouldn’t be treated unfairly or lose your job because you ‘blow the whistle’.
You can raise your concern at any time about an incident that happened in the past, is happening now, or you believe will happen in the near future.
Who is protected by law
You’re protected if you’re a worker, eg you’re:
- an employee, such as a police officer, NHS employee, office worker, factory worker
- a trainee, such as a student nurse
- an agency worker
- a member of a Limited Liability Partnership (LLP)
Get independent advice if you’re not sure you’re protected, e.g. from Citizens’ Advice.
A confidentiality clause or ‘gagging clause’ in a settlement agreement isn’t valid if you’re a whistleblower.
Complaints that count as whistleblowing
You’re protected by law if you report any of the following:
- a criminal offence, eg fraud
- someone’s health and safety is in danger
- risk or actual damage to the environment
- a miscarriage of justice
- the company is breaking the law, e.g. doesn’t have the right insurance
- you believe someone is covering up wrongdoing
News 19th October
Chain reaction – the publication of the Leeds data has triggered fresh revelations from ex-employees of Hygiene Solutions. Apparently the Leeds machines have been set at 5% for several years. This implies that thousands of Deprox “terminal cleans” have been done at St James’s Hospital in Leeds, in which the machine would have produced no measurable disinfection or reduction in pathogens whatever. Countless vulnerable patients will have been put in to rooms that are still dangerously contaminated. This is criminal. The fraudulent Deprox system needs to be purged out of NHS hospitals immediately.
News 13th October

Dr Nick Brown. Hygiene Solutions “man on the inside” at Addenbrooke’s.
Information received yesterday sheds light on the relationship between Hygiene Solutions Ltd boss Rick Fentiman and Addenbrooke’s Consultant Microbiologist Dr Nick Brown.
Dr. Brown has been closely associated with Hygiene Solutions for many years, and his contact details are given to prospective customers as a prime “reference” to confirm the efficacy of the Deprox system. Dr Brown’s study at Addenbrooke’s Hospital, published in 2012, has been made in to a PowerPoint presentation and is used by Deprox salesman Tom Lister to promote the product.
This study relates to a “bundle” of interventions apparently including Deprox, that were implemented between 2002 and 2009. However, after the report was published in the Journal of Hospital Infection, a response in the form of a letter from Phillipe Destrez was published, pointing out that the hydrogen peroxide decontamination system in use at Addenbrooke’s in this period was actually the Glosair 400 system by Advanced Sterilization Products. Phillipe pointed out that Hygiene Solutions was only registered as a company in 2010. The Deprox trademark and patent were registered in 2012.
In fact, Dr. Nick Brown gave a presentation of this particular Addenbrooke’s study at the Hospital Infection Society 2010 conference in Liverpool, accrediting Sterinis (previous name of Glosair) as having been used at this time.
Dr Brown has some questions to answer as to why his published paper credited Deprox, when he was clearly aware that this was incorrect.
It is doubtless just a coincidence that Hygiene Solutions financed what must have been a very expensive research project for Dr Brown. – see below. The email correspondence between Rick Fentiman and Nick Brown is very revealing – Rick’s advice to Tom Lister, his Deprox salesman to be “aware and cautious on this one” is practically an admission of guilt.
The second report, which was financed by Hygiene Solutions, was published in 2013.
www.publish.csiro.au/view/journals/dsp_journal_fulltext.cfm?nid=241&f=HI13009
The report summarised the results of 1840 surface contact plate samples taken from 58 rooms in Addenbrooke’s before and after the Deprox process. These showed the average efficacy of Deprox to be less than log2 – four orders of magnitude less than claimed. These results are not particularly surprising, as competing systems that work on the same principle of aerosolising a 5% solution only achieve a 2-3log kill.
It is very odd that a report entirely by UK authors, financed by a UK business and based on research at a UK hospital should only be published as a letter in an obscure Australian journal (which has now been discontinued). Was this an attempt to suppress the dismal Deprox performance results, while still allowing the authors to add one more “published paper” to their scoreboards?
News 12th October
From newly “released” data, an up to date list of the locations of all Deprox units in existence. Much more data to follow, watch this space…
| Country/City | Hospital | Deprox Units |
| ENGLAND | ||
| Andover | Andover War Memorial Hospital | 1 |
| Basingstoke | Basingstoke and N. Hants Hospital | 4 |
| Bradford | Bradford Royal Infirmary | 1 |
| Bury St Edmunds | West Suffolk Hospital | 1 |
| Cambridge | Addenbrooke’s Hospital | 11 |
| Cambridge | Papworth Hospital | 1 |
| Canterbury | Kent and Canterbury Hospital | 1 |
| Cannock | Cannock Hospital | 1 |
| Colchester | Colchester General Hospital | 4 |
| Croydon | Croydon University Hospital | 1 |
| Dudley | Russells Hall Hospital | 3 |
| Ealing | Ealing Hospital | 1 |
| Fleet | Fleet Community Hospital | 1 |
| Halifax | Calderdale Royal Hospital | 2 |
| Harrow | Northwick Park Hospital | 1 |
| Huddersfield | Huddersfield Royal Infirmary | 1 |
| Kettering | Kettering General Hospital | 2 |
| King’s Lynn | On call decontamination service units at depot. | 35 |
| Leeds | St James’s Hospital | 8 |
| Leeds | Leeds General Infirmary | 2 |
| Leicester | Leicester General Hospital | 1 |
| Leicester | Leicester Royal Infirmary | 1 |
| Leicester | Glenfield Hospital | 1 |
| Liverpool | Royal Liverpool Hospital | 5 |
| Liverpool | The Walton Centre | 1 |
| Liverpool | Broadgreen Hospital | 1 |
| London | UCLH (Now reallocated) | 4 |
| Luton | Luton and Dunstable Hospital | 1 |
| Manchester | Royal Oldham Hospital | 1 |
| Manchester | Manchester Royal Infirmary | 1 |
| Manchester | Wythenshawe Hospital | 2 |
| Manchester | North Manchester General Hospital | 1 |
| Mansfield | Kings Mill Hospital | 2 |
| Margate | QEQM Hospital | 2 |
| Reading | Royal Berkshire Hospital | 2 |
| Redditch | Alexandra Hospital | 2 |
| Sheffield | Northern General Hospital | 4 |
| Torquay | Torbay Hospital | 1 |
| Walsall | Walsall Manor Hospital | 1 |
| Winchester | Royal Hampshire County Hospital | 5 |
| Wolverhampton | New Cross Hospital | 4 |
| Worcester | Worcester Royal Hospital | 1 |
| WALES | ||
| Abergavenny | Neville Hall Hospital | 1 |
| Bridgend | Princess of Wales Hospital | 2 |
| Newport | St Woloos/ Ysbyty Ystrad Fawr | 1 |
| Newport | Royal Gwent Hospital | 2 |
| Port Talbot | Neath Port Talbot Hospital | 2 |
| Rhyl | Glan Clywd Hospital | 1 |
| Swansea | Singleton Hospital | 4 |
| Swansea | Morriston Hospital | 2 |
| AUSTRALIA | ||
| Sydney | Westmead Hospital | 20 |
| Sydney | Concord Hospital | 1 |
| Sydney | Mater Hospital | 2 |
News 10th October
A mass of new information has just come in to my hands – current location and service history of each of the 183 Deprox machine in existence, including documentary proof that the St James’s Hospital (Leeds) Deprox units with serial numbers 050, 051 and 053 have been turned down to ΔRH 5%. All this information will be published shortly – watch this space…
Meanwhile, hits on deproxfraud.info soared past 7500 this evening, with renewed interest from New Zealand following the closure of the Auckland office.
News: 7th October
Gallant Maxwell, the Soho solicitors who are vainly trying to defend fraudsters Hygiene Solutions Ltd are hardly above reproach themselves. A Google search of the company immediately pops up their listing on SOLICITORS FROM HELL. The AVOIDING BAD SOLICITORS SITE has awarded them three lame ducks. Now they have some more quacks to deal with…
News: 5th October
When did you last test your Deprox? The Health and Social Care Act 2008 requires that healthcare providers VALIDATE decontamination equipment and ENSURE THAT DECONTAMINATION PROCESSES ARE FIT FOR PURPOSE. The Deprox system can be simply and inexpensively tested by using off-the-shelf BIs (Biological Indicators). Why take a risk? Ask your hospital’s microbiologists to do a quick test.


News: 4th October
- Gallant Maxwell, Hygiene Solutions’ lawyer, threatens site host WordPress with legal action. WordPress confirm that they will not be removing the site. Simon Gallant says ” The business will just go down the toilet, I’m afraid…”
- Congratulations to Deprox salesman Tom Lister, whose speech at the Infection Prevention 2016 Conference in Harrogate has been viewed 400times as of this afternoon.
News: 3rd October
- The Australian distributor of the Deprox system pulled out, and deleted the product from their website:

 http://acutehealthcare.com.au/products/http://acutehealthcare.com.au/products/environmental-decontamination/
http://acutehealthcare.com.au/products/http://acutehealthcare.com.au/products/environmental-decontamination/
- Addenbrooke’s Hospital in Cambridge uses more Deprox units than any other hospital in the world. When Deprox was introduced to the UCLH hospital, infection rates soared, see “UCLH Tragedy” This morning, the Daily Mail reports a superbug outbreak in the Neonatal Intensive Care unit at Addenbrooke’s.
News: 2nd October
An update from inside the Hygiene Solutions office.
- Staff report that the directors seem very tense, pale and preoccupied, and spend much of their time huddled in the board room with various serious looking strangers.
- The deproxfraud.info website has been blacklisted and can no longer be accessed from staff computers, rather pointlessly as they have phones…
- Staff have been issued with instructions on how to respond to phonecalls relating to the fraud, and are to tell customers that “legal action” is being taken against the site’s author.
- Plans are afoot to partition off “Aisle G” so it is not visible to D&H customers.
- All staff have been put on high alert as to approaches by strangers, including the warning that they may be being videotaped at any time.
Deproxfraud Goes Viral!
 Deproxfraud.info has gone viral! 3081 views in just 2 days have resulted from a car park leafleting campaign at the Infection Prevention conference in Harrogate on September 28th. As only 300 cars were targeted, and probably less than half of these belonged to the Infection Control delegates (There were 2 other exhibitions running at the same time) these figures can only be explained by healthcare professionals spreading the word in the hospital community. Just another virus that Hygiene Solutions can’t kill…
Deproxfraud.info has gone viral! 3081 views in just 2 days have resulted from a car park leafleting campaign at the Infection Prevention conference in Harrogate on September 28th. As only 300 cars were targeted, and probably less than half of these belonged to the Infection Control delegates (There were 2 other exhibitions running at the same time) these figures can only be explained by healthcare professionals spreading the word in the hospital community. Just another virus that Hygiene Solutions can’t kill…