Hygiene Solutions claim a single Deprox unit has the capacity to decontaminate rooms with a volume of 380m3, e.g. a 12 bed ward bay. A typical hospital side ward (single room with ensuite) has a volume of 60m3.
However, the chamber used by Hygiene Solutions Ltd to test the Deprox is 1.5m x 1.5m x 2.8m. Total volume 6.3m3 , just 10% of the volume of a hospital single bedroom, and 1.7% of the maximum volume that Deprox is guaranteed to disinfect. It is barely larger than a telephone box.
Hygiene Solutions internal testing, published here for the first time reveals that the Deprox, in spite of being boosted with 50% more concentrated hydrogen peroxide solution than the standard “Deproxin” was incapable of a log 6 decontamination of even this tiny test chamber.
The Deprox was thoroughly tested over a period of months by David Sempere Aracil, a well qualified chemist. David placed Log 6 biological indicator discs (Apex Biological Indicator #HMV-091) in 8 different locations around the inside walls of the test chamber. The Deprox unit (“Trusted by leading hospitals around the world”) was sealed in the chamber, and the process was run. The log6 BIs were incubated – they were all alive.
David tried substituting Sanosil SO15 which at 7.5% H2O2 is 50% more concentrated than Deproxin. Now some of the BIs would be sterilised, sometimes. Over several weeks in late 2014, David did a series of 12 tests in the test chamber, all with 7.5% H2O2 rather than the 5% Deproxin. He tried turning the ΔRH up and down, but to no avail. In 5 of these tests, all 8 BIs remained viable. None of the tests sterilised more than 6 out of 8.
In summary then:
Deprox, running on a 5% H2O2 solution, is claimed to give a log6 decontamination of an entire 380m3 ward, including inside small crevices and complex equipment. In Hygiene Solutions’ own tests, the Deprox running on a 7.5% solution, and thus generating a 50% higher aerial H2O2 concentration than the standard process, completely failed to give a log6 decontamination of a 6.3m3 box in multiple tests.
Hygiene Solutions continued to promote and sell the Deprox with exactly the same claims, but in 2015, they turned the whole Deprox fleet down from ΔRH20 to ΔRH5. See https://deproxfraud.info/2017/03/13/leaked-emails-prove-test-cheating-bodily-harm-and-massive-fraud/
Fortunately, (or unfortunately for Hygiene Solutions) David’s notes of these tests survived.
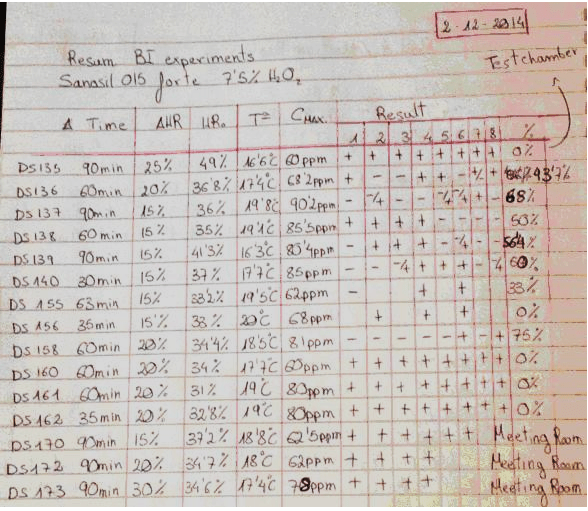
Explanation of table.
This table is a summary of 15 tests done by David Sempere Aracil, assisted by Tautvydas Karitonas, over a period of months. Both are university graduates with extensive research experience, and David has a PhD in Chemistry. The tests were done with a standard production model Deprox machine, the purpose of the tests was to determine if the extremely low efficacy of the Deprox process could be rectified by increasing the concentration of the hydrogen peroxide solution from the standard 5% to 7.5%.
The results were recorded in 3 A4 hardcover notebooks. Each of the 15 tests was recorded in more detail on preceding pages of the notebook. In addition to the efficacy tests, the notebooks contain extensive details of tests on prototype catalyst systems, and constitute proof that HS was well aware of both the low efficacy and residual gas issues with Deprox.
Heading: “Sanosil 015 forte” refers to Sanosil S015, which is a disinfectant intended for water systems. It is 7.5% Hydrogen peroxide solution. Note that this is more concentrated than the 5% Deproxin solution that is used in production Deprox machines.
Col.1. The test number. These are not sequential, as some tests did not use Biological indicators (BIs) and were not recorded in this resume.
Col. 2 Duration of test measured from when the machine starts vapourising. (It takes several minutes for the machine to fill the piezo tank at the beginning of each test)
Col. 3 Delta HR setting of machine. This is adjusted by using unmarked pressure sensitive switches below the LCD display. – see How to test your Deprox.
Col. 4 HRO This is the original relative humidity in the test chamber before the machine starts.
Col. 5 TO Temperature in the chamber before the machine starts
Col. 6 CMAX Hydrogen peroxide concentration in PPM, maximum level reached during process.
Results columns. The first 12 tests were done in the test chamber (wardrobe). Each number represents a specific marked location on the test chamber wall where an exposed stainless steel Bacillus subtilis log6 biological indicator was placed. The chamber is a crude wood and plasterboard structure in an essentially unheated warehouse. It is approximately 5’ x 5’ x 9’ and the indicators were placed at various heights on the interior walls of the chamber. The last 3 tests were done in the company board room which is approximately 12’ x 25’.
A” +” indicates that the BI still contained viable bacteria, a “–“ indicates that all bacteria on this indicator were killed.
Final column. This is the percentage of BIs that were killed.








 The effect of this is to give heavy droplet wetting on the upwards facing surfaces around the machine. Bed rails, for example will be wet on the top and dry on the bottom. This has the fortuitous (for the manufacturer) effect, that if the machine is tested in the customary manner by placing biological indicators around the room in petri dishes facing upwards these samples will be exposed to a substantial “rain” of droplets and may show high efficacy levels which are not at all representative of the real disinfection achieved. If the BIs were secured in a vertical or downwards facing orientation, much lower efficacies would be recorded.
The effect of this is to give heavy droplet wetting on the upwards facing surfaces around the machine. Bed rails, for example will be wet on the top and dry on the bottom. This has the fortuitous (for the manufacturer) effect, that if the machine is tested in the customary manner by placing biological indicators around the room in petri dishes facing upwards these samples will be exposed to a substantial “rain” of droplets and may show high efficacy levels which are not at all representative of the real disinfection achieved. If the BIs were secured in a vertical or downwards facing orientation, much lower efficacies would be recorded.