Ultra-V and Spectrome are Hygiene Solutions’ attempt to penetrate the UV room disinfection market by using the same underhand and dishonest tactics as they used to promote the fraudulent Deprox device. As with Deprox, the data presented on their website is rudimentary – the more controversial claims are made in tender responses and sales presentations out of the public gaze, where they are less likely to be challenged.
UV systems are very simple and hence there is little to choose between different manufacturers. From a marketing perspective, if a system can claim some unique and novel advantage, it can displace the competition and also sell for a higher price.
Ultra-V is apparently the only UV system that can disinfect shadowed areas! A hospital in the west of England that was approached by the company and told that Ultra-V will achieve a 4-6 kill log reduction of pathogens in “shadowed areas, the underside of beds, behind units and in hidden corners.” (Fortunately, the hospital “smelt a rat” and cancelled the tender…)
Indeed, this is what the Ultra-V brochure says:

There is nothing unusual or novel about the “Ultra-V”. It has ten standard 5′ germicidal UV tubes, where more powerful competing systems may have up to 24. In fact, it is very similar to the American made Tru-D system on which the design was based.
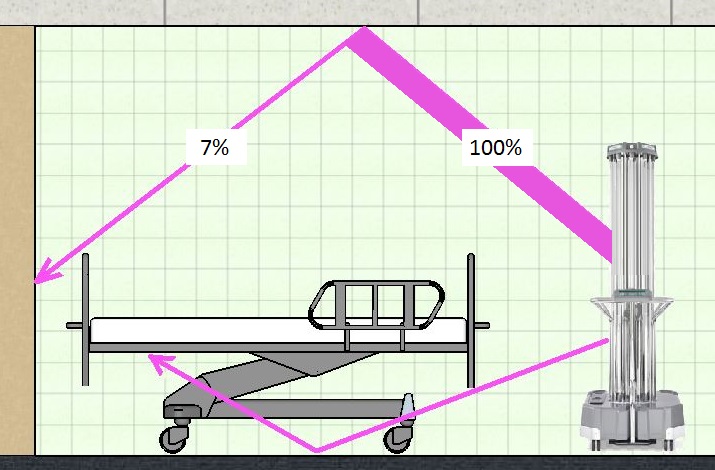
The basis for the “shadowed areas” claim is that the system comes with a set of detachable “Spectromes” which are crude light meters that are in Bluetooth contact with the main unit. These meters are distributed around the room, including in shadowed areas, and the theory is that the process time is extended, if necessary, until the shadowed (i.e. indirectly lit) areas have received the full 4 to 6 log dose of UV light. Sure enough, if a “Spectrome” is placed in a shadow, the process time extends from 15 to as much as 40 minutes to give the extra dose.
The problem is this:
UV-C light is strongly absorbed by almost all floor wall and ceiling materials and paints likely to be found in a hospital room – far more so than visible light. A study of UV-C disinfection systems in hospital rooms found that the UV-C reflectivity of standard paint was between 5% and 7%. In other words, if we could see the world in UV-C, the walls, floor and ceiling would appear to be essentially black.
The only UV light that reaches shadowed areas, i.e. areas not in direct line of sight of the UV tubes, is reflected, scattered light. These reflected rays have lost over 90% of their power, and as log efficacy of UV disinfection is linear with exposure time, the lamps will have to run for at least 10 times as long if the shadowed areas are to receive a full dose of UV radiation. In other words, if 15 minutes gives a log 4-6 reduction for directly exposed surfaces, the “shadow treatment” process as claimed for Ultra-V will take about 2 ½ hours.
This estimate is confirmed by the results of the study mentioned above, using a Tru-D machine, which is almost identical to the Ultra-V. The table below was extracted from the paper.

Here the unit is run for nearly 44 minutes in a 130 ft² room. (i.e. approx 11′ x 11′) The average log reduction of the 33 samples that were directly illuminated is log 3.41. The average log reduction of the 27 samples in the shadows was only log 2.01.
How then…does the Ultra-V achieve log4-6, in the shadows, in about 30 minutes?
The answer is very simple – it doesn’t. Regardless of what the shadowed “Spectromes” may demand, the system is set to a maximum run time of 40 minutes or so – because otherwise customers would complain. (The rapid process time is perhaps the greatest “selling point” of UV as compared to alternative processes. A system that ran for 2 hours or more would be uncompetitive, and would also damage and bleach materials in the treated area by overexposure.) However, for the hospital’s peace of mind, the machine will automatically generate and email a report stating that the room has been cleaned to log 4 standards.
Yes, but… Hygiene Solutions tested the efficacy with contact plates when the system was trialled, and the results were excellent!
The nice man from Hygiene Solutions is indeed very willing to do all the microbiological testing free of charge. To quote from a UK hospital that conducted a trial of the system:
” There was personnel on site from the company throughout the trial, the engineer conducted sampling and used 5 contact plate samples in each area the unit was used. These samples were then sealed, labelled, logged with start and finish times and area sampled, these were then refrigerated in the company vehicle before being transported to their own laboratory for testing.”
I have no direct information for the Ultra-V testing, but former Deprox operators have reported that the Biological Indicators that they brought back from “validating” their Deprox processes were put on the sales rep’s desk and remained there indefinitely. There is little evidence that they were ever incubated or cultivated at all. Could it be that the graduate microbiologist at Hygiene Solutions can determine the results by intuition, without resorting to the old fashioned and time consuming process of incubating the samples?
However, if we assume that the plates are incubated and the results honestly reported, what does that tell us? Almost nothing, according to the following article by Michrochem Laboratory, who are the leading independent UV system testing lab in the USA.
“Unfortunately, environmental swab studies are confounded by several problematic technical factors, described in detail below. Taken together, these factors make environmental swab studies some of the least reliable means of testing UV effectiveness.”
“The first major confounding factor of environmental swab studies on UV efficacy determinations is mathematical in nature. Initial microbial populations in indoor or hospital environments are often low. There are are frequently only about 100 total bacteria per 10 square centimeters of surface. That is not much of a challenge for many UV systems, meaning the extent of the UV effects may not be fully measurable. On top of that, lab techniques used to enumerate microorganisms on the swabs often result in a poor limit of detection, meaning that viable cells on the surface may not be detected if they are present in low numbers.”
“The second problematic aspect of environmental swab studies is related to microbiological technique. Populations of microorganisms often vary widely from one spot to the next, even on the same surface. If the same exact location were swabbed before and after treatment this would not be a problem, but the act of swabbing a surface or sampling it with a press-plate effectively cleans the surface, removing microorganisms in the process. Swabbing pressure and surface area are also variable. Even the best researchers find it challenging to swab different surfaces, yet maintain the same pressure and cover the same surface area. Doorknobs and sink handles, for example, are more challenging than a table section.”
The 65mm RODAC contact plates used by Hygiene Solutions have an area of 33cm². From the data above, a typical bacterium density is 10 CFU/cm², so the “before” plate might be expected to show about 330 CFU. In order to demonstrate a log4 reduction, it is obvious that the plate would have to show at least 10,000 CFU.
To compound this, the contact plate obviously removes the bacteria from the patch tested. Hence almost inevitably, regardless of whether any treatment has taken place or not, a second plate applied to the same patch will have a lower colony count – because the first plate effectively cleans the surface!
The only way to prove a log4-6 reduction is to use a mixture of log4 and log6 biological indicators in both the shadowed and unshadowed areas. As C. difficle spores are much more resistant to UV than vegetative organisms, and are also one of the most common and threatening of the serious pathogens, these should be the test organism of choice. Because of the inherent conflict of interest in a decontamination equipment supplier doing its own testing, an independent test laboratory, or the hospital’s own microbiology department should do the testing. If the equipment supplier is reluctant to agree to this, or offers to take the samples and “have them tested by an independent lab” – walk away!
Yes but…there are published papers in highly regarded medical journals that demonstrate the efficacy of the Ultra-V. So it must work as claimed!
The Journal of Hospital Infection published the following article in July 2016.

A single sentence sums up the results of this study:

Touch plate tests were done before and after a manual clean, then after a 20 minute Ultra-V cycle. There is no suggestion that shadowed areas were tested. The reduction attributable to the Ultra-V is therefore a reduction from 19.3 CFU to 2.3 CFU. This is a little less than a ten fold reduction, i.e. it is less than log 1.
Just to remind ourselves, Hygiene Solutions claim a log4-6 reduction even in shadowed areas. That is a reduction of between 10,000 and 1,000,000 fold.
The actual reduction achieved was 8 fold – rather less than log 1.
Court order re. false and misleading statements
Interestingly, Ultra-V’s alter ego, the Tru-D device is also in trouble…
Sadly, the Ultra-V fraud has human consequences – as Hygiene Solutions point out in their brochures, some of their customers are using this shockingly inadequate log 1 system to clean high risk areas such as operating theatres.

Finally, to add insult to injury, Hygiene Solutions are trying to sell this contraption, which is essentially a standard fluorescent light unit on wheels, for an astonishing £50,000+
Comments:


















Nelson Bridwell
So what is the solution? Multiple smaller light sources around the room? Reflective wall paint? A mobile light source that drives around the room, following a stripe on the floor?